IR technology is a rapid and convenient tool for both qualitative and quantitative analysis that has been around for over a century. Traditional IR spectroscopy relies on vibration energies from the molecular bindings, where IR emission is absorbed by the bond when it has the same frequency as the specific vibration or movement as the bond.
This specific absorption wavelength (nm) (or wavenumber, cm-1, as is frequently used in the world of IR spectroscopy) can be used for the determination of the sum of hydrocarbons in water samples. They first “just” need to be transferred to a suitable solvent, free of bonds, that show absorption in the same range.
Looking at parameters such as oil and grease, and total petroleum hydrocarbons (TPH) that utilize IR detection, there are two main approaches. Each are covered by an ASTM method.
In ASTM D7066-04, Standard Test Method for dimer/trimer of chlorotrifluoroethylene (S-316) Recoverable Oil and Grease and Nonpolar Material by Infrared Determination, the IR absorption coming from the –CH bond asymmetric stretch vibration at around 2930 cm-1 is measured. I.e. this requires a solvent free of CH-bonds. The ASTM method stipulates S-316 solvent to be used, however, an alternative good solvent for extraction that is free of CH-bonds is tetrachloroethylene, which has been tested successfully on the Biotage® Horizon 5000.
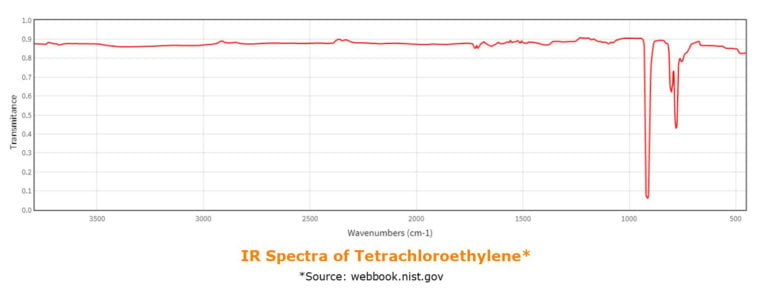
The alternative approach, used in ASTM D7678 – 17 Standard Test Method for Total Oil and Grease (TOG) and Total Petroleum Hydrocarbons (TPH) in Water and Wastewater with Solvent Extraction using Mid-IR Laser Spectroscopy utilizes the -CH3 bend absorption, that shows IR absorption in the range of 1370-1380 cm-1. As cycloalkanes do not have any terminal CH3 groups they are excellent non-chlorinated alternatives at this wavenumber.
Well, as always, when things are looking too good there should always be a pitfall. Whereas IR detection at 2930 cm-1 easily can be run on your standard FT-IR instrument using a simple cuvette holder and a fairly cheap NIR quartz cuvette, detection a 1370-1380 cm-1 demands quite a bit more of the optical pathway. The liquid cell or low cell needs to be made of special material such as zinc selenide (ZnSe) or arsenic selenide (As2Se3). However, there are instruments designed purely for this type of analysis, if the non-chlorinated path is preferred.
Up to now, both these principles have relied on liquid-liquid extraction (LLE) of the water sample prior to analysis. Now, Biotage can offer a straightforward automated process for the extraction part to reduce labor, decrease the exposure risk for solvents, and minimize error sources, etc., using the Biotage® Horizon 5000 instrument and Pacific® Premium Oil & Grease Disks, for extraction of the samples. Both principles have been tested, using either tetrachloroethylene or cyclohexane.
When running these methods, there are some important considerations to take into account. Both methanol and acetone are used in the traditional oil and grease/TPH analytical protocols, and yes, both of these have -CH / CH3 bounds, so some care needs to be taken to avoid interference in the final analysis.
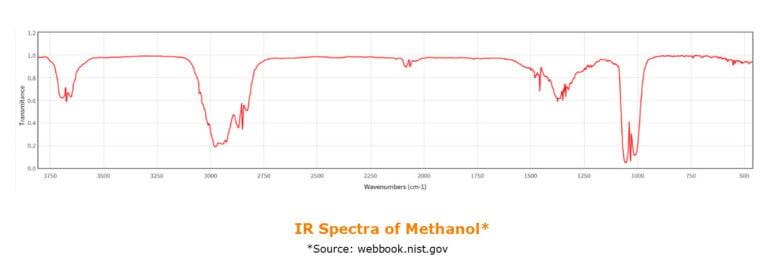
Traditionally the disk, as in this case the Pacific Premium O&G disks are conditioned with methanol prior to loading the samples. In applications where FT-IR detection has been used, methanol pre-wet was simply omitted and a quick pre-rinse with respective solvent was successfully used before processing the aqueous sample.
The spiking solution for quality control samples are often prepared in acetone. However, acetone must be removed before the elution using the IR suitable solution. This was easily done by just adding a simple rinse step with reagent water just after the sample has been loaded.
With these two considerations in mind, with a focus on what the final detection is actually measuring and two simple solutions to avoid interference, the door is opened for a new – but century-old – detection technique for an important application field for the Biotage® Horizon 5000 instrument.
Read more on solid phase extraction below.

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership