Correct pretreatment of samples prior to supported liquid extraction (SLE) produces better partitioning and cleaner extracts. Supported liquid extraction (SLE) is a simple clean-up technique similar to liquid-liquid extraction (LLE), but on a solid supported surface.
SLE is an easy, affordable sample prep technique similar to liquid-liquid extraction. The best way to do an SLE+ extraction is discussed here. You typically pretreat the sample to adjust the pH, load onto the SLE+ cartridge, wait 5 minutes, and elute with a water immiscible organic solvent. Analytes partition from the aqueous sample into the organic solvent. Analytes must be hydrophobic enough to prefer the organic solvent to the aqueous environment or they will be retained on the SLE+ sorbent. If the aqueous sample is at a pH where the compounds of interest are charged, that can affect extraction efficiency. Ideally, analytes you want to extract using SLE+ should be neutral in solution. The more polar the analyte, the more important neutralization is.
Using the pH rule of 2 (see figure 1, below), choose a pretreatment buffer that will result in your compounds being non-ionized. For basic analytes, you want to pretreat with something at least 2 units above the pKa, for acids you want your sample to be at least two units below the pKa. Two good sources of pKa data are chemicalize.com and the human metabolome database. If you have a panel of acidic or basic drugs, choose a pretreatment two units higher than the highest pKa for a panel of basic drugs and 2 units below the lowest pKa for an acidic drug panel. 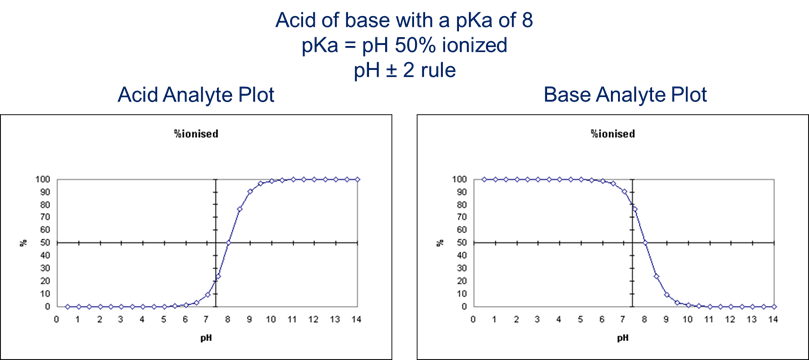
If you have a large panel of drugs, you can adjust the pH so that the majority of analytes are neutral, or you may tailor the pretreatment to maximize recovery of the more polar analytes. If you have very hydrophobic compounds with higher logP values, like 9-carboxy-THC, they may partition into your SLE extraction solvent even if they are charged. However, more polar, hydrophilic compounds, or compounds with a negative logP will extract better if they are neutral. If you’re not sure, try acidic, neutral and basic pH pretreatments with several elution solvents, and determine recovery, matrix effect and process efficiency to determine the best conditions. There is a great chart on page 14 of the ISOLUTE SLE+ User Guide with recommended buffers and elution solvents for SLE method development.
If you are working with serum or plasma and your analytes are bound to plasma proteins, try pretreating your plasma or serum 1:1 with 2% formic acid or 50:50 isopropanol:water to disrupt plasma proteins and release bound analytes. Underloading the SLE+ by 10-25% results in better partitioning and cleaner extracts when organic solvent like isopropanol is used. A load volume of 300-350 µL on a 400 µL ISOLUTE SLE+ cartridge is a good starting point. For whole blood samples, pretreat with base for basic drugs, or lyse with water using sonication or centrifugation to lyse the cells. Some analytes, like immunosuppressants, are bound to red blood cells and lysing is required to release the analytes. Hydrolyzed urine samples can be pretreated with acid, base, water or neutral buffer depending upon the compound properties.
For more information on designing your sample prep method development experiments watch the webinar ““Demystifying Sample Prep - A Systematic Approach to Sample Prep Method Development for Clinical, Forensic and CRO Laboratories” below.

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership