In the world of environmental testing, we all know that contamination happens. It can be frustrating trying to locate where it is coming from when you are also trying to keep up with the samples that continue to come through the door.
I know that narrowing down the specific location of contamination within your workflow can be quite a lengthy process, so I will walk you through each step to ensure that we are looking at one variable at a time and remedy the contamination in a timely manner. Let’s look at how to pinpoint contamination in three essential pieces of the extraction workflow for Gas Chromatography Mass-Spectrometry (GC-MS) analysis.
Analytical Instrument
First you want to make sure that your instrument blank is passing, meaning no contamination of target analytes present. There are a lot of EPA methods the require you to run an instrument blank anyways, but even if it isn’t required, it could be highly beneficial. They can determine if there’s carryover from your high-level calibration curves points, your continuing calibration verification (CCV) standard, sample carryover, as well as simple instrument background. You can inject an instrument blank into your sequence wherever you would like! If you see contamination present within your instrument blanks, then you’ll want to give your instrument a good clean-up with fresh supplies.
Another thing that you want to make sure to check is that your continuing calibration verification (CCV) is passing your instruments performance criteria. If your instrument isn’t functioning properly and not recovering the check standard properly, then samples will need to be re-ran. The CCV helps to ensure the analytic instrument is providing optimal performance. If your GC-MS is not performing properly, then it will be nearly impossible to track down the source of your laboratory contamination!
Extraction Solvents
If your instrument blanks are clean, then you’ll want to check your extraction solvents. Calculate the typical volumes of each solvent used to extract your samples. Place the full volume of fresh extraction solvents into your concentration instrument and concentrate the solvent using your normal parameters. Prepare the final solvent volume for analysis on the analytical instrument.
You might be asking yourself, “how is that different from an instrument blank?” Well, an instrument blank consists of 1 mL of solvent (usually). By conducting this experiment, for example, you’re concentrating 50 mL of solvent down to 1 mL. You’re effectively testing the amount of contamination present within 50 mL of solvent instead of 1 mL, increasing the level of possible contamination by 50. This will be more representative of the amount of contamination that is introduced when using the full volume of extraction solvents within your procedure.
If you find that there’s contamination here, then you’ll want to double check the grade of solvent that you’re using. Try another vendor or a higher grade of solvent and complete the same test and see if the contamination is still present.
Extract Drying Agents
Another common location for contamination is your extract drying agents. To test this step, follow the same preparation procedure as the extraction solvents section above. Now, have your mock extract undergo your normal drying procedure. Concentrate your dried extract following your normal parameters and once complete, analyze on your instrument.
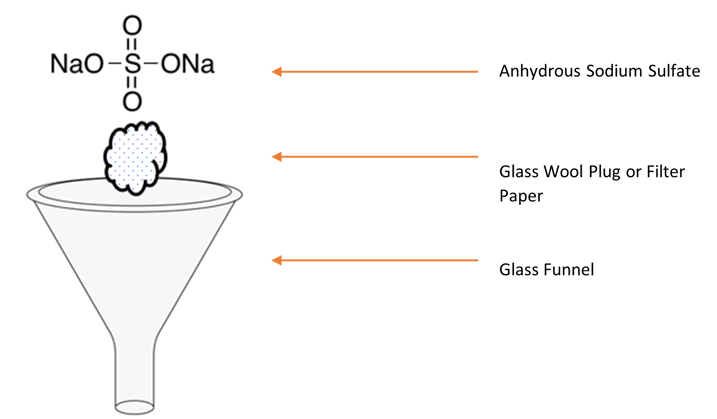
If you notice contamination here, then it could be from the glass wool, filter paper, sodium sulfate, or solvent phase separation paper (if applicable). If you are using sodium sulfate, ensure that your filter paper or glass wool is clean as well as the sodium sulfate. Please note: some glass wool could be treated, so you want to make sure that it is baked and cleaned before you use it. Filter paper may require a quick rinse to waste once it is placed in the funnel, so be sure to do that as well. If you are using solvent phase separation paper, then ensure that the paper and apparatus is also cleaned in preparation of drying your extract. Repeat the drying step with another mock extract, concentrate, and analyze.

 Organic Workflow
Organic Workflow Peptide Workflow
Peptide Workflow Scale-Up Flash Purification
Scale-Up Flash Purification  Sample Preparation
Sample Preparation Biomolecule Purification
Biomolecule Purification Oligo synthesis
Oligo synthesis Scavengers and Reagents
Scavengers and Reagents Service & Support
Service & Support Accessories & Spare parts
Accessories & Spare parts Investors
Investors Reports & News
Reports & News The Share
The Share Corporate Governance
Corporate Governance Calendar
Calendar Sustainability
Sustainability Our Offering
Our Offering Our History
Our History Our Locations
Our Locations Leadership
Leadership